When winter arrives, the drastic reduction in the driving range of electric vehicles becomes one of the most frustrating pain points for countless EV owners. What kind of transformation would it bring to our lives if a battery could not only operate stably in the freezing cold of –50 °C but also maintain high efficiency in the scorching heat of 50 °C?
Recently, a team led by Professor Xueliang Sun, Chair Professor at the Eastern Institute of Technology, Ningbo (EIT) and Foreign Member of the Chinese Academy of Engineering, and Associate Professor Xiaona Li of the College of Engineering at EIT, along with their collaborators, has proposed a brand-new solution to realize this vision.
Addressing one of the key challenges in next-generation solid-state battery technology — the inherent incompatibility between halide solid-state electrolytes and the lithium metal anode — the team creatively devised an interface design strategy of turning an adversary into an ally. By exploiting the thermodynamic instability between the solid-state electrolytes and lithium metal, they precisely tuned the electrolytes composition to guide the in situ growth of a favorable solid electrolyte interface (SEI), rendering all-solid-state lithium metal batteries with outstanding comprehensive performance. The related research findings were published in the Journal of the American Chemical Society, a top chemistry journal, under the title "Design for an Interface in Oxyhalide-Based All-Solid-State Lithium Metal Batteries."
The "Achilles' Heel" of Solid-State Batteries
All-solid-state lithium metal batteries offer prominent advantages of high safety and theoretical energy density. However, the interfacial incompatibility between the solid-state electrolytes and the lithium metal anode remains the core bottleneck restricting their performance.
Take the high-performance oxyhalide solid-state electrolyte as an example. It possesses high ionic conductivity, but its interface compatibility with the lithium metal anode is extremely poor. It is readily reduced by lithium to form an interface that is electronically conductive yet ionically insulating. This subsequently triggers continuous side reactions, exacerbates interfacial impedance, and induces a risk of lithium dendrite penetration, ultimately leading to short circuits.
The current conventional approach is to insert an "isolation wall" (such as an indium foil or sulfide buffer layer) in between. This not only complicates the battery structure but also introduces additional interfacial resistance. Therefore, abandoning the buffer layer to achieve the direct and stable compatibility of oxyhalide electrolytes with the lithium metal anode, and enabling robust operation under high current density, high areal capacity, and a wide temperature range, has become a critical breakthrough point for the practical application of solid-state lithium metal batteries.
Turning an Adversary into an Ally: Spontaneous Interface Regulation
Faced with this challenge, the team took a contrarian approach — since the reaction is unavoidable, why not make it work in our favor?
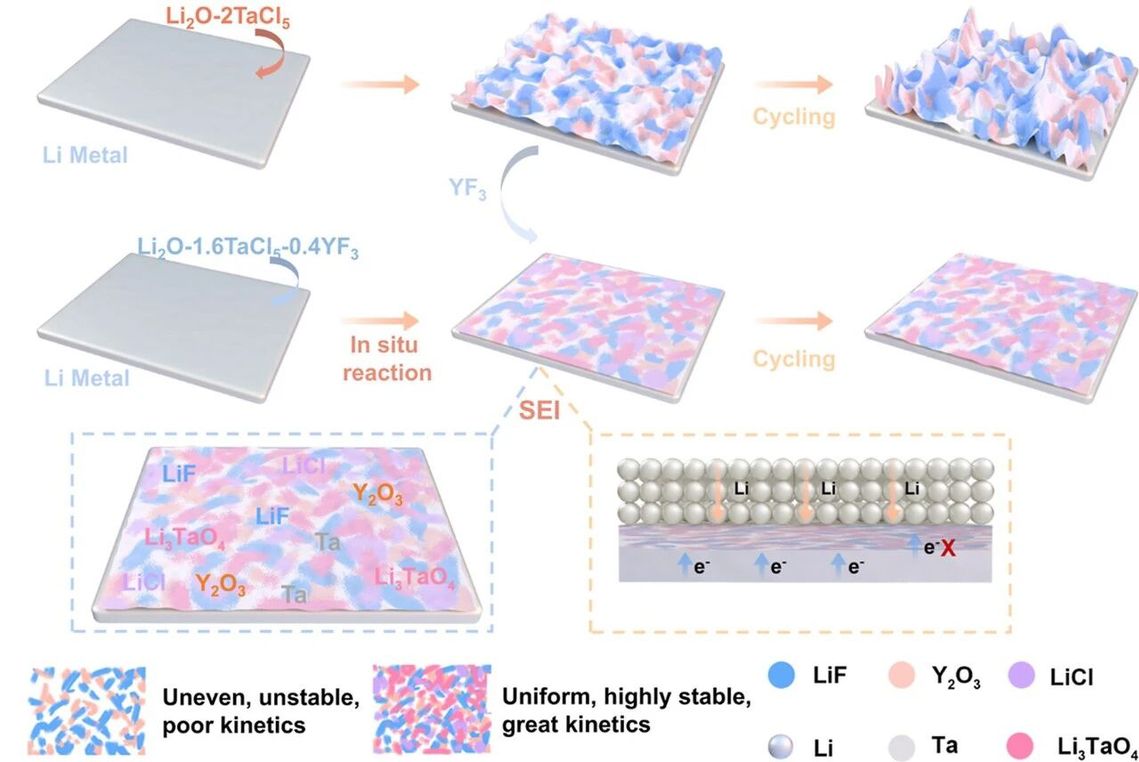
Schematic illustration of the spontaneously induced surface-interface functional layer
By doping the oxyhalide electrolyte with yttrium fluoride (YF₃), the research team skillfully altered the reaction pathway. Upon contact between the electrolyte and lithium metal, a composite solid electrolyte interphase (SEI) composed of multiple inorganic species — including lithium chloride (LiCl), lithium fluoride (LiF), and yttrium oxide (Y₂O₃) — is spontaneously formed in situ. This interphase acts like a tailor-made armor; it prevents electrons from continuing to wreak havoc while allowing lithium ions to pass through efficiently, fundamentally resolving the issues of interfacial degradation and lithium dendrite formation.
From Laboratory to Application: Stunning Performance
Based on this innovative strategy, the all-solid-state batteries constructed by the team delivered a stunning report card:
Ultra-long lifespan: At a current density of 10 mA cm⁻², the Li||Li symmetric cell can cycle stably for over 11,000 hours, demonstrating robust interfacial stability.
Exceptional tolerance: The battery achieves a critical current density as high as 12.7 mA cm⁻², implying excellent fast-charging potential.
Ultra-wide temperature range: The all-solid-state battery can operate stably over a temperature range of –50 °C to 50 °C, promising to overcome the chronic ailment of batteries being averse to both cold and heat.
Excellent compatibility: This system can be directly paired with commercial nickel-cobalt-manganese (NCM) ternary cathodes and high-voltage lithium cobalt oxide (LiCoO₂) cathodes, eliminating the need for additional coating modifications on the cathode material. This greatly simplifies the preparation process of all-solid-state batteries and reduces costs.
Dr. Yanlong Wu, Ph.D., at China Automotive Battery Research Institute, and faculty member at the School of Mathematics and Science, Nanyang Institute of Technology, is the first author. Dr. Jianwen Liang, Director of GRINM (Guangdong) Institute for Advanced Materials and Technology, and the National Power Battery Innovation Center of China Automotive Battery Research Institute; Dr. Jiantao Wang, Deputy General Manager of China Automotive Battery Research Institute; Professor Xueliang Sun, Chair Professor at EIT and Foreign Member of the Chinese Academy of Engineering; and Associate Professor Xiaona Li at EIT, are the corresponding authors.
This research was supported by the Beijing Natural Science Foundation, the Ningbo Yongjiang Talent Introduction Program, the National Natural Science Foundation of China, the National Youth Talent Support Program and the Beijing Nova Program, among others.