All-solid-state batteries are hailed as the ultimate form of next-generation batteries, yet high costs and suboptimal energy density remain significant hurdles to their industrialization.

Future all-solid-state battery model (AI-generated)
A team led by Professor Xueliang Sun, Chair Professor at the Eastern Institute of Technology, Ningbo and Foreign Member of the Chinese Academy of Engineering, and Professor Changhong Wang, Vice Director of the Institute of Matter and Energy at the Eastern Institute of Technology, Ningbo, in collaboration with partners, has achieved new progress in the field of halide solid electrolytes. They report for the first time a low-cost, high-ionic-conductivity (2.55 mS/cm) oxyhalide solid electrolyte with asynchronous activity. The findings were recently published in the renowned international journal Advanced Materials.
Research Background
All-solid-state batteries are considered a crucial direction for next-generation rechargeable batteries due to their high energy density and intrinsic safety. The key to the current industrialization of all-solid-state batteries lies in developing solid-state electrolytes that combine high ionic conductivity, a wide electrochemical stability window, and low cost. Halide electrolytes have garnered significant attention for their high room-temperature ionic conductivity and strong oxidation resistance, but they often rely on rare metals such as tantalum (Ta) and gadolinium (Gd), leading to high costs and relatively high electrochemical inactivity in composite cathodes.
To reduce costs, research has shifted towards halide systems containing abundant elements like zirconium (Zr), aluminum (Al), and iron (Fe). Although the material cost of these systems can be reduced to below $50/kg, their ionic conductivity typically remains around 1 mS/cm, and they require excessive proportion in the composite cathode, limiting cell-level energy density. Recently proposed redox-active halide electrolytes, which combine lithium-ion conduction and energy storage functions, still face challenges such as insufficient conductivity or reliance on expensive metals.
The core challenge addressed by this research is: how to achieve solid-state electrolytes that are simultaneously cost-effective, high-performing, and capable of enhancing battery energy storage capacity.
Research Highlights
1. Low-Cost, High-Ionic-Conductivity, High-Activity Oxyhalide Solid-State Electrolyte
This paper reports for the first time a novel, highly efficient active oxyhalide solid electrolyte, exhibiting a high ionic conductivity of 2.55 mS/cm and a reversible capacity of 163 mAh/g.
Compared to Li₂ZrCl₆, the LiZrFeOCl-1604 oxyhalide solid electrolyte achieves a 15.3% reduction in material cost, a 5.8-fold increase in ionic conductivity, and a 12.1% decrease in activation energy. Furthermore, by embedding Fe-based redox chemistry in the LiZrFeOCl-1604 oxyhalide solid electrolyte, a significant reversible capacity of 163 mAh/g is obtained.
2. Asynchronous Activation Mechanism Significantly Enhances Energy Density of Composite Cathodes
The study discovered that an asynchronous activation mechanism can enhance the energy density of composite cathodes. This mechanism enables asynchronous iron redox activity, providing additional capacity when coupled with LiFePO₄ (LFP) cathode material. The synthesized active oxyhalide solid electrolyte functions as a lithium-ion conductor within the voltage range of LFP while simultaneously offering additional reversible redox capacity beyond this voltage window. Consequently, the composite electrode demonstrates a high discharge capacity of 321.6 mAh/g, an energy density of 982.1 Wh/kg (based on LFP mass), and excellent cycling stability exceeding 800 cycles at a 1C rate. These performance metrics significantly surpass those of electrochemically inert Li₂ZrCl₆ and other reported active halide solid-state electrolytes.
3. Novel Synthesis Approach for Active Oxyhalide Solid Electrolytes
This study also proposes a novel synthesis strategy for active oxyhalide solid electrolytes. By combining inexpensive iron oxide (FeOₓ) metal oxide precursors with a non-stoichiometric synthesis route, an amorphous oxyhalide solid-state electrolyte was successfully prepared using a one-step mechanical ball milling method. This provides a highly cost-effective alternative to the previously reported amorphization method for Li₂ZrCl₆.
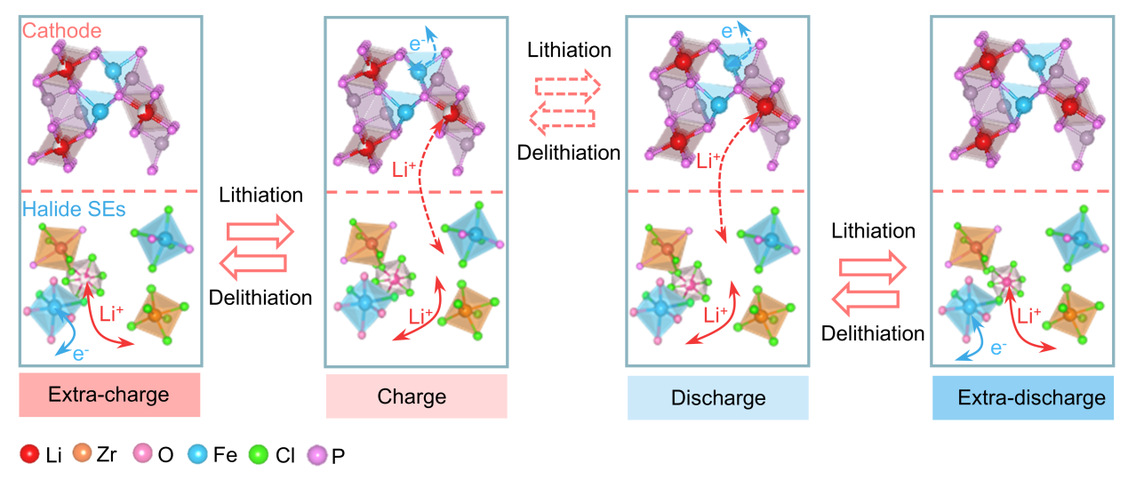
Schematic of the charging/discharging process in a composite cathode utilizing the asynchronously active oxyhalide solid-state electrolyte
The Eastern Institute of Technology, Ningbo is the first affiliation of this paper. Professor Xueliang Sun and Changhong Hong, and Professor Liwei Chen from the School of Chemistry and Chemical Engineering at Shanghai Jiao Tong University are the corresponding authors. Zhimin Zhou and Pushun Lu are the co-first authors.
Link: https://doi.org/10.1002/adma.72694