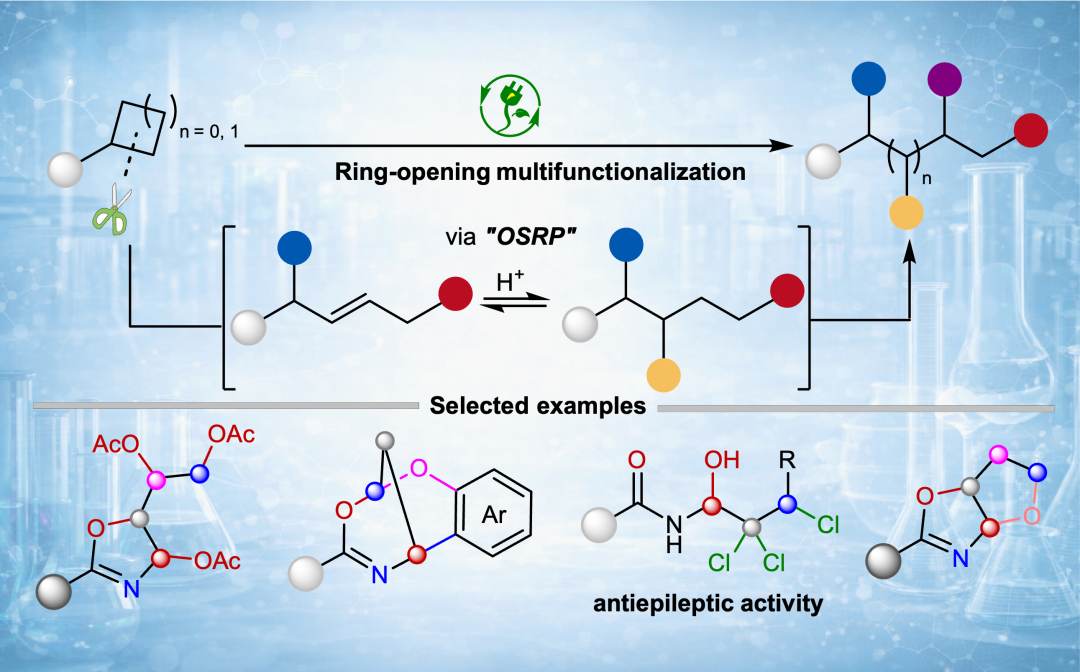
In the world of chemistry, strained rings such as cyclopropanes and cyclobutanes behave like compressed springs, storing considerable reaction driving force. Yet a long-standing problem has remained: after ring-opening, reactions typically stop at the 1,3- or 1,4-difunctionalization stage, making it extremely difficult to continue "adding" functional groups on adjacent carbons.
Now, a collaborative team led by Associate Professor Chen Zhu of Eastern Institute of Technology, Ningbo and Associate Professor Tao Shen of Shanghai Jiao Tong University has developed a programmable, divergent electrochemical strategy that achieves, for the first time, multi‑site ring‑opening multifunctionalization of strained rings. For future drug and functional molecule synthesis, this means chemists can start from simpler strained rings and more efficiently "grow" complex structures that were previously inaccessible in one step.
The results were recently published in Nature Chemistry.
Why is adding just one more step so difficult?
Strained ring opening is a common tool for building complex molecules. The problem is that after the initial functionalization, the reactivity of the surrounding C-H bonds often drops significantly. Controlling site, regio and oxidation state among multiple inert C-H/C-C bonds simultaneously is extremely challenging. Yet continuous C-O, C-N and Carbon-halogen skeletons are widespread in natural products, drug molecules and high-value intermediates. Enabling strained rings to go from "one opening" to "sequential, precise editing" - giving chemists a tool that can "take several more steps" - has been a central challenge in the field.
Ring-opening multifunctionalization of strained rings. Image provided by the research group
Installing a "throttle valve" on a highly reactive intermediate
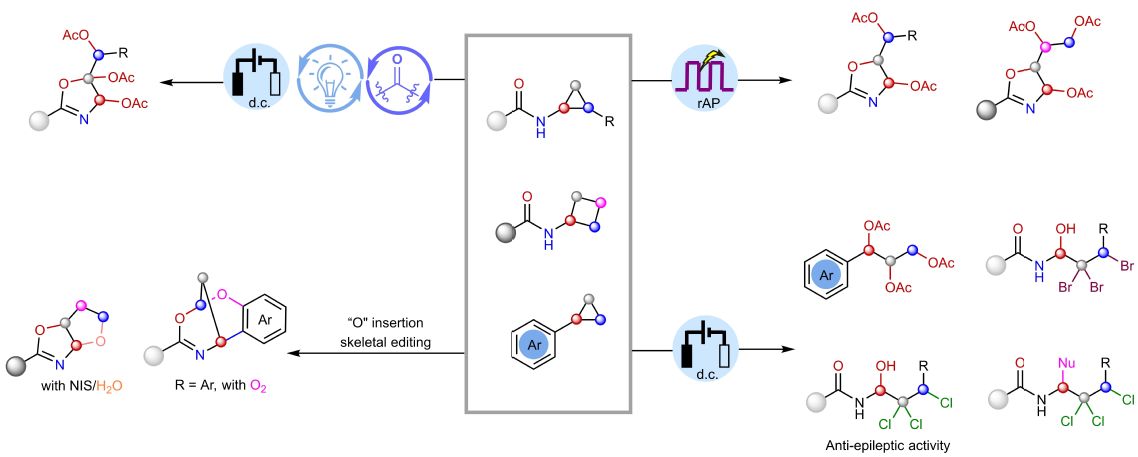
The team proposed an elegant concept: the "Olefin slow‑release pool" (OSRP).
In simple terms, after the strained ring opens for the first time, it does not undergo uncontrolled oxidation. Instead, under acidic conditions, it slowly and reversibly releases an olefin intermediate, and subsequent oxidation and functional group installation occur stepwise under electrochemical conditions.
This acts like a "throttle valve" on a highly reactive intermediate:
It prevents olefin polymerization and over oxidation.
It creates a "selectivity window" for sequential activation of C-C/C-H bonds.
For the first time, multi-step oxidation becomes controllable.
Growing diverse high-value molecules from the same platform
Based on this concept, the team integrated direct‑current (d.c.) electrolysis, rapid alternating polarity (rAP) electrolysis and electrophotocatalysis into a single programmable platform. By tuning the acid, the anode potential and the electrolysis mode, they could selectively achieve trioxygenation, tetraoxygenation, trihalohydroxylation and "O-insertion" skeletal editing from the same class of strained ring substrates. Simple, readily available cyclopropanes and cyclobutanes were directly converted into high-value structures such as oxazolines, polyols, polyhalogenated alcohols, as well as bicyclic frameworks via skeletal editing.
The method works for both aryl- and alkyl-substituted substrates with different substitution patterns, is compatible with drug-relevant fragments, and has been demonstrated on a gram scale- showing practical potential.
DFT calculations reveal the reaction roadmap
In this work, Chen Zhu's group carried out the key Density functional theory (DFT)calculations and mechanistic studies.
The calculations show that:
The reaction first undergoes anodic oxidation to generate a radical cation intermediate, which significantly lengthens and weakens the C-C bond of the strained ring, promoting ring opening.
This is followed by an intramolecular hydrogen-atom transfer to form the key olefin intermediate in the OSRP. This pathway has a lower energy barrier than direct elimination (23.9 vs 37.9 kcal/mol), which explains why the reaction is more controllable.
For the cyclopropaneamides system, the calculations further reveal that under photosensitized conditions, hydrogen-atom transfer occurs preferentially at the α-C-H bond adjacent to oxygen- exactly the reason why the tetraoxygenated product is built with high regioselectivity.
The cyclobutaneamide system requires a higher potential, which is also consistent with the calculated higher free energy change for oxidation. Together, these results outline a unified mechanistic framework for the oxidative ring‑opening multifunctionalization of cyclopropanes and cyclobutanes.
From difunctionalization to multifunctionalization
This work moves strained ring opening chemistry from the traditional difunctionalization into a new phase of programmable, multi-site, multi-oxidation-state control.
It not only provides a new tool for the rapid construction of highly oxidized complex molecules, but also highlights the unique advantages of electrochemistry in the precise activation of inert C-C/C-H bonds.
The first author of the paper is Yajuan Li, a Ph.D. student at Shanghai Jiao Tong University. Dr. Yatao Lang, a postdoctoral fellow at Eastern Institute of Technology, Ningbo, contributed to the DFT calculations and mechanistic studies. Associate Professor Chen Zhu of Eastern Institute of Technology, Ningbo, and Associate Professor Tao Shen of Shanghai Jiao Tong University are the co-corresponding authors. This work was supported by grants from the National Natural Science Foundation of China, the Fundamental Research Funds for the Central Universities, the Shanghai Jiao Tong University 2030 Initiative, the Shanghai Qiyuan Innovation Foundation, the National Science and Technology Major Project, and the Fujian Provincial Natural Science Foundation, among others.